History of genetic testing
In order to understand the genetic tests used in the diagnosis of Turner syndrome, it is important to understand the genetic changes that lead to the condition. A chromosome is a structure that organizes the genetic material in the body. Usually, humans have 23 pairs of chromosomes (46 total). Two of these chromosomes (X and Y) determine biological sex. Females typically have two X chromosomes and males have one X and one Y. However, genetic changes can alter the number or structure of chromosomes. Most females diagnosed with Turner syndrome are missing one copy of the X chromosome. However, structural changes to the X chromosome or mosaicism can also cause this condition (Gravholt et al., 2023). The following paragraphs provide a brief overview of the genetic tests that can be used in the diagnosis of Turner syndrome. The tests will then be described in more detail.
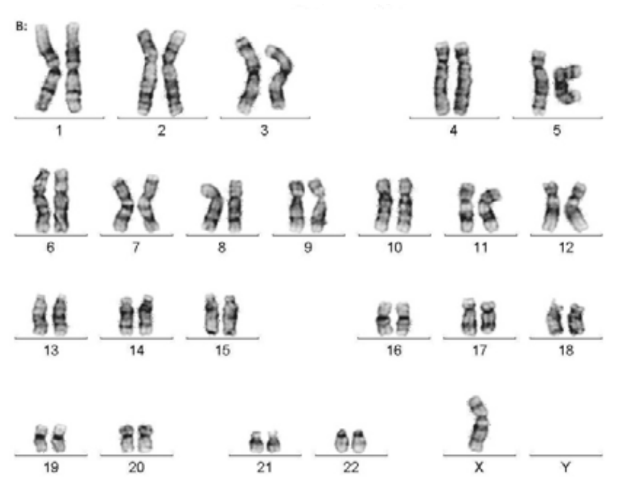
Image 1: Visual representation of chromosomes - missing one copy of X
(Filho, 2013)
In 1959, Dr. Charles Ford determined that Turner syndrome was caused by a genetic anomaly using a technique called karyotyping (Gravholt et al., 2023). This testing method allows laboratory technicians to visualize all of the chromosomes (usually 23 pairs) in an individual’s cells. In the late 1980s, fluorescence in situ hybridization (FISH) was introduced into clinical practice to identify chromosome anomalies. In the context of Turner syndrome, this method involves using a fluorescent probe to identify changes to the X chromosome (Cui et al., 2016). Chromosomal microarray (CMA) was introduced a few years after FISH in the early 2000s. This technique is also effective for detecting altered copies of the X chromosome (Levy & Wapner, 2018).
Currently, these are the most common genetic tests used in the diagnosis of Turner syndrome. Each test has specific advantages which will be described in the following sections of this post. All three tests can be performed once the laboratory receives a sample from the patient. Typically, a blood sample is used. However, other tissues can be examined for analysis (Gravholt et al., 2023).
Prenatal diagnosis of Turner syndrome is more complex, because physicians cannot perform a standard blood draw on babies in the womb. However, prenatal screening and prenatal diagnostic testing has improved over the last decade. It is important to recognize the differences between these two categories of testing. A screening test can assess the likelihood of Turner syndrome, but it cannot provide a definitive answer as to whether or not the baby will have the genetic condition (Carlson & Vora, 2017). Conversely, a diagnostic test can provide a definitive answer as to whether or not the baby will have Turner syndrome. Diagnostic testing requires the pregnant person to undergo an invasive procedure so genetic material can be retrieved from the fetus or the placenta (Carlson & Vora, 2017). Since these procedures are invasive, there is a risk of miscarriage. Once a sample is obtained from a diagnostic procedure, it will be sent to the laboratory and technicians will perform karyotyping, FISH, or CMA to evaluate the baby for Turner syndrome.
How is genetic testing for Turner syndrome more advanced today?
As stated above, karyotyping, FISH, and CMA are standard techniques for identifying Turner syndrome in childhood and adulthood. The same tests are used to detect Turner syndrome prenatally if a sample is retrieved through a diagnostic procedure. However there has been a significant advancement in prenatal screening for Turner syndrome. In 2011, a technique called prenatal cell-free DNA (pcfDNA) screening was introduced into clinical practice to screen for chromosome anomalies such as Turner syndrome (Carlson & Vora, 2017). Pregnant patients often undergo subsequent diagnostic testing if the pcfDNA screening results suggest that there is a high likelihood that the baby could be affected by Turner syndrome. There are two main types of prenatal diagnostic testing, chorionic villus sampling and amniocentesis (Carlson & Vora, 2017).
Before pcfDNA was available, there were not many effective screening options for Turner syndrome. Historically, specific ultrasound findings have been associated with the condition. These ultrasound markers are still considered relevant today, and they include increased fluid at the back of the neck, specific heart defects such as coarctation of the aorta, and shortened limbs (Chen & Chien, 2007). It is important to note that these ultrasound findings can also be indicative of other chromosome anomalies. Therefore, when these markers are observed on ultrasound, prenatal screening and/or diagnostic testing is often recommended to determine a specific diagnosis.
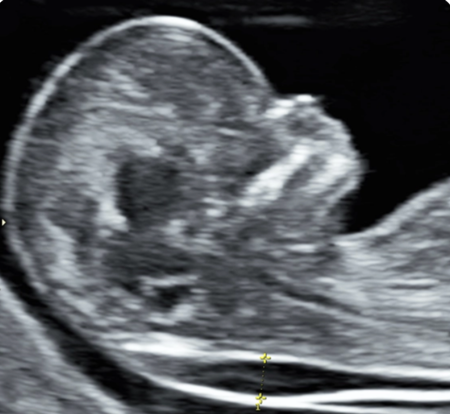
Image 2: Ultrasound showing a baby with increased fluid at the back of the neck
(London Pregnancy Clinic, 2024)
What does each test tell you?
Karyotype
A karyotype allows for the visualization of chromosomes. This technique can be used to determine if one copy of the X chromosome is absent. It can also identify large structural differences of the X chromosome (Gravholt et al., 2023).
Fluorescence in situ hybridization (FISH)
FISH uses fluorescent probes to identify the presence or absence of genetic material. Like karyotyping, FISH is able to detect the absence of the X chromosome and large structural changes. Unlike karyotyping, FISH is able to detect small structural differences (Cui et al., 2016).
Chromosomal microarray (CMA)
Similar to karyotyping and FISH, chromosomal microarray can determine whether a patient has both copies of the X chromosome. Like FISH, CMA can identify large and small structural changes. CMA is usually capable of detecting changes that FISH is unable to identify (Levy & Wapner, 2018).
Prenatal cell-free DNA (pcfdna)
As stated above, pcfDNA is a prenatal screening test. Throughout pregnancy, fragments of placental cells are present in mom’s bloodstream. To perform a pcfDNA test, a blood sample is taken from mom and the laboratory analyzes the placental fragments within the sample (Carlson & Vora, 2017). Typically, the genetics of the placenta reflect the genetics of the fetus. Therefore, pcfDNA can be used to estimate the likelihood that a baby will be born with Turner syndrome. PcfDNA is a non-invasive test, but there is no increased risk to the fetus and it can be performed once the fetus is 10 weeks old (Carlson & Vora, 2017).
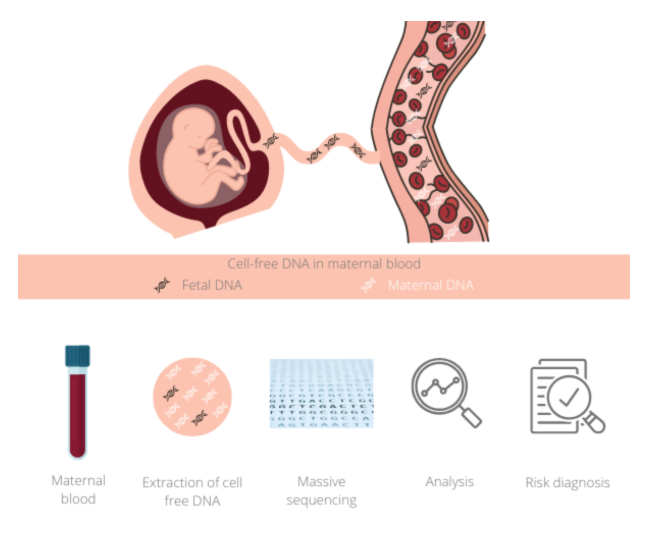
Image 3: Visual representation of pcfDNA screening
(Genosalut, 2022)
Chorionic villus sampling (CVS)
CVS is one type of prenatal diagnostic testing that is usually performed between 10 to 14 weeks of gestation. This is an invasive test that retrieves a sample from the chorionic villi of the placenta (Schulman & Elias, 1993). Chorionic villi are structures that connect the placenta to the uterus. This procedure can be performed through the abdomen or the cervix, depending on provider preference. Due to the fact that this is an invasive test, it can lead to miscarriage (Schulman & Elias, 1993). The number quoted for the risk of miscarriage varies between medical centers. In 2014, an article published in the journal of Ultrasound Obstetrics and Gynecology suggested that the procedure-related risk of CVS is approximately 0.11% (Akolekar et al., 2014). Once a sample of chorionic villi is retrieved through CVS, the tissue can be sent to the laboratory for karyotyping, FISH, or CMA.
Amniocentesis
Amniocentesis is another type of prenatal diagnostic testing that is typically performed between 16 to 20 weeks gestation. This is an invasive test that is performed through the abdomen to retrieve amniotic fluid which consists of fetal skin cells and urine (Schulman & Elias, 1993). Since this is an invasive procedure, it can lead to miscarriage. Like CVS, the number quoted for risk of miscarriage varies between medical centers. In 2014, the same article that reported on the procedure related risk of CVS suggested that the procedure related risk of amniocentesis is 0.22% (Akolekar et al., 2014). After amniotic fluid is retrieved through amniocentesis, the sample can be sent to the laboratory for karyotyping, FISH, or CMA.
How Do You Get a Genetic Test?
Genetic testing can be ordered once a patient has had an appointment with a medical geneticist or a genetic counselor. These health care professionals can help patients determine the most appropriate testing method. Patients can be referred to genetic specialists by any medical doctor if they believe that genetic testing is warranted. A doctor will typically refer someone to genetics if a specific genetic condition runs in the patient’s family, if the patient has not met developmental or growth milestones, or if they have unexplained infertility (Chappelle et al., 2009). Pregnant patients are always referred to genetics by maternal fetal medicine providers or gynecologists if they are 35 or older. This is due to the fact that there is a slightly higher risk for chromosome anomalies at age 35. Pregnant patients can also be referred to genetics specialists if any of their routine ultrasounds show findings that are consistent with a genetic condition (Chappelle et al., 2009).

Image 4: Speaking with a genetic counselor
(Sanmati Genetic Counseling Center, 2022)
References
Akolekar, R., Beta, J., Picciarelli, G., Ogilvie, C., & D’Antonio, F. (2015). Procedure-related risk of miscarriage following amniocentesis and chorionic villus sampling: A systematic review and meta- analysis. Ultrasound in Obstetrics & Gynecology, 45(1), 16–26. https://doi.org/10.1002/uog.14636
Best Genetic Counseling Services you can trust. Sanmati Genetic Counseling Center. (2024, January 29). https://www.sgccenter.com/
Carlson, L. M., & Vora, N. L. (2017). Prenatal Diagnosis. Obstetrics and Gynecology Clinics of North America, 44(2), 245–256. https://doi.org/10.1016/j.ogc.2017.02.004
Chappelle, A., Terry , S., Hawkins, A., Garrison, A., Christensen, K., & Burke, B. (2009). Understanding genetics: A New York, Mid-Atlantic guide for patients and Health Professionals. Genetic Alliance.
Chen, C. P., & Chien, S. C. (2007). Prenatal Sonographic Features of Turner Syndrome. Journal of Medical Ultrasound, 15(4), 251–257. https://doi.org/10.1016/S0929-6441(08)60042-9
Cui, C., Shu, W., & Li, P. (2016). Fluorescence In situ Hybridization: Cell-Based Genetic Diagnostic and Research Applications. Frontiers in Cell and Developmental Biology, 4. https://doi.org/10.3389 /fcell.2016.00089
Gravholt, C. H., Viuff, M., Just, J., Sandahl, K., Brun, S., Van Der Velden, J., Andersen, N. H., & Skakkebaek, A. (2023). The Changing Face of Turner Syndrome. Endocrine Reviews, 44(1), 33–69. https://doi.org/10.1210/endrev/bnac016
Fetal DNA test in maternal blood. Genosalut. (2022, July 6). https://www.genosalut.com/en/news/fertility-and-pregnancy/cell-free-fetal-dna-test/
Filho, H. (2013, February 13). Applicability of data mining technique using Bayesians network in diagnosis of genetic diseases. Academia.edu. https://www.academia.edu/2568935/APPLICABILITY_OF_DATA_MINING_TECHNIQUE_USING_BAYESIANS_NETWORK_IN_DIAGNOSIS_OF_GENETIC_DISEASES
Levy, B., & Wapner, R. (2018). Prenatal diagnosis by chromosomal microarray analysis. Fertility and Sterility, 109(2), 201–212. https://doi.org/10.1016/j.fertnstert.2018.01.005
Increased nuchal translucency. London Pregnancy Clinic. (2024, June 26). https://www.londonpregnancy.com/nuchal-translucency-increased-risk/
Schulman, L., & Elias, S. (1993). Amniocentesis and Chorionic Villus Sampling. The Western Journal of Medicine, 159(3), 260-268.
Written by Haley Stambaugh, TSF volunteer blog researcher/writer. Edited and designed by Kesha Amin, TSF blog coordinator.
© Turner Syndrome Foundation, 2025




